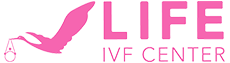Many people assume that embryo transfer is simply the final checkbox in IVF, a quick clinical moment after all the “real” work is done. That assumption can actually set patients up for disappointment. Embryo transfer is, in fact, the pivotal point where science meets biology, where months of preparation either succeed or stall. Understanding exactly what happens during this procedure, what influences its outcome, and how to prepare effectively can meaningfully change your perspective on fertility treatment and strengthen your partnership with your care team.
Table of Contents
- What is embryo transfer? Core concepts and definitions
- How embryo transfer works: The procedure step by step
- The importance of uterine preparation: Fresh vs frozen embryo transfers
- Success rates and what affects embryo transfer outcomes
- Personalizing your path: Key questions to ask and next steps
- What most people miss about embryo transfer—and what actually works
- Explore advanced embryo transfer support with Life IVF Center
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Embryo transfer defined | Embryo transfer is the pivotal IVF step where embryos are placed into the uterus to achieve pregnancy. |
| Preparation matters most | Optimizing the uterine lining with medication and timing increases the chance of implantation success. |
| Success rates vary | Factors like age, diagnosis, and embryo quality greatly influence individual outcomes beyond national averages. |
| Ask the right questions | Knowing what to discuss with your fertility team can personalize your treatment and improve decision-making. |
What is embryo transfer? Core concepts and definitions
Embryo transfer is defined clearly in reproductive medicine: as a step in assisted reproduction where an embryo is placed into the uterus with the intent to establish a pregnancy. It is used most commonly as part of IVF (in vitro fertilization), but it also applies to other ART (assisted reproductive technology) methods, including embryo donation and gestational carrier cycles.
Within IVF specifically, embryo transfer is the culmination of a carefully sequenced process. To understand why it matters so much, it helps to see where it sits in the overall timeline.
Where embryo transfer falls in the IVF timeline:
- Ovarian stimulation: Fertility medications stimulate multiple follicles to develop.
- Egg retrieval: Mature eggs are collected from the ovaries under sedation.
- Fertilization: Eggs are fertilized in the laboratory with sperm, creating embryos. You can learn more about this phase when exploring fertilization and embryo culturing.
- Embryo development: Embryos are cultured for three to five days, reaching blastocyst stage in many cases.
- Embryo transfer: One or more embryos are placed directly into the uterus.
- Pregnancy testing: A blood test is conducted approximately ten to fourteen days after transfer.
Reviewing what to expect in IVF at each stage helps patients feel less overwhelmed and more in control of their journey.
“Precise placement of the embryo within the uterine cavity is not incidental. Research consistently links transfer technique and positioning accuracy to implantation rates, making the skill of the clinical team a genuine variable in IVF outcomes.”
This step is often misunderstood because it appears simple on the surface. Patients sometimes focus all their anxiety on egg retrieval, which involves sedation and physical recovery. The transfer, by contrast, takes only a few minutes and requires no anesthesia in most cases. Yet it is the moment that determines whether a pregnancy begins.
With a foundational understanding of its relevance, let’s move to what actually happens during the embryo transfer itself.
How embryo transfer works: The procedure step by step
The procedure is precise, brief, and designed to minimize discomfort. If you are preparing for IVF or approaching a transfer cycle, knowing what to expect clinically can ease a great deal of anticipatory stress.
The embryo transfer process, step by step:
- Bladder preparation: Patients are typically asked to arrive with a moderately full bladder. This helps ultrasound imaging produce a clearer view of the uterus during the procedure.
- Positioning: You lie on an examination table in a position similar to a routine gynecological exam.
- Speculum insertion: A speculum is gently inserted to allow the physician to visualize the cervix clearly.
- Catheter placement: A thin, flexible transfer catheter is passed through the cervical canal and into the uterine cavity. This is the most critical technical moment of the procedure.
- Embryo loading: A laboratory embryologist loads the embryo (or embryos) into the catheter with a tiny amount of culture media.
- Embryo deposition: Using ultrasound guidance, the physician deposits the embryo at a precisely targeted location within the uterus, typically in the upper third of the uterine cavity.
- Catheter removal and confirmation: The catheter is carefully withdrawn and examined under a microscope by the embryologist to confirm the embryo has been fully released.
- Rest period: Patients rest briefly before being discharged, usually within thirty to sixty minutes of arrival.
Anesthesia is generally not required, and most patients describe the sensation as mild pressure or light cramping. The entire in-room procedure takes roughly five to ten minutes, though preparation and recovery extend the clinic visit.

Pro Tip: Ask your physician ahead of time about their preferred catheter type and whether they routinely use ultrasound guidance. Studies suggest ultrasound-guided transfers result in higher clinical pregnancy rates compared to clinical touch alone, and clinics that consistently use this approach often demonstrate stronger outcomes.
Embryo transfer is also a central step in third-party IVF options, including cycles that use donated eggs or gestational carriers. In those cases, the recipient’s uterine preparation takes on heightened importance, which brings us to the next critical topic.
The importance of uterine preparation: Fresh vs frozen embryo transfers
Embryo transfer success depends on far more than the physical act of placing an embryo. The uterine lining, called the endometrium, must be in an optimal state of receptivity. Timing this window correctly is one of the most clinically nuanced aspects of reproductive medicine.
Uterine receptivity and endometrial preparation are particularly critical in frozen embryo transfers, where patients receive estrogen and progesterone to coordinate the implantation window. This hormonal coordination is carefully calibrated based on each patient’s individual cycle characteristics.
Fresh vs. frozen embryo transfer: Key differences
| Feature | Fresh embryo transfer | Frozen embryo transfer (FET) |
|---|---|---|
| Timing | 3 to 5 days after egg retrieval | Weeks to months after retrieval |
| Uterine preparation | Natural cycle post-retrieval | Hormone-medicated protocol |
| Medications required | Fewer hormonal supplements | Estrogen and progesterone regimens |
| Scheduling flexibility | Limited; tied to retrieval cycle | Greater control over transfer timing |
| Risk of ovarian hyperstimulation | Higher (OHSS concern) | Lower; ovaries have recovered |
| Common use case | Fresh donor egg cycles | Banked embryos; genetic testing results |
Frozen embryo transfers have increased significantly in recent years, partly because preimplantation genetic testing (PGT), which screens embryos for chromosomal abnormalities, requires time to process results. Freezing allows the uterus to recover from stimulation medications before transfer, which many specialists believe creates a more receptive environment. Details on cryopreservation and protocols can help patients understand how embryos are safely stored and prepared for future use.
Pro Tip: Endometrial thickness is regularly monitored via ultrasound before a frozen transfer. A lining thinner than 7 millimeters is often associated with lower implantation rates. If your lining is consistently thin, ask your physician about extended estrogen protocols or adjunct therapies that may improve receptivity before proceeding.
Hormone protocols for frozen transfers typically begin with oral or transdermal estrogen to thicken the uterine lining, followed by the introduction of progesterone to trigger the implantation window. The precision of this timing is what separates a well-prepared transfer from a premature one.
With a sense of how preparation matters, the next logical concern for couples is: what are realistic embryo transfer success rates, and what influences these outcomes?
Success rates and what affects embryo transfer outcomes
Success rates are one of the first things patients search for, and one of the most commonly misread pieces of information in reproductive medicine. In the United States, fertility clinics are required to report outcomes to the CDC (Centers for Disease Control and Prevention) through the SART (Society for Assisted Reproductive Technology) data system. This reporting framework creates a standardized basis for comparison, but it also has important limitations.
ART success rates are reported by clinics to the CDC using clinic-specific and national data, and outcomes vary by factors such as age and diagnosis. Notably, the “own eggs” category includes embryo transfers that occur within one year after an egg retrieval, meaning single-cycle and banked-embryo outcomes are grouped together.
Typical live birth rates by age group and egg source
| Age group | Own eggs (estimated live birth rate per transfer) | Donor eggs (estimated live birth rate per transfer) |
|---|---|---|
| Under 35 | 45% to 50% | 50% to 55% |
| 35 to 37 | 35% to 42% | 50% to 54% |
| 38 to 40 | 25% to 32% | 49% to 53% |
| 41 to 42 | 15% to 20% | 48% to 52% |
| Over 42 | Under 10% | 47% to 51% |
Ranges are approximate national averages. Individual clinic data and patient-specific factors vary considerably.
What clinics and national averages cannot tell you:
The CDC emphasizes that average success rates may not represent outcomes for a specific couple’s circumstances. A 45% national average for women under 35 tells you very little about your individual odds if you have diminished ovarian reserve, endometriosis, or a prior failed transfer.
Factors that significantly affect embryo transfer success:
- Patient age: Egg quality declines with age, which directly impacts embryo quality and implantation potential.
- Embryo quality: Blastocyst-stage embryos, particularly those with normal chromosomal profiles after PGT, have higher implantation rates.
- Uterine health: Conditions such as fibroids, polyps, or thin endometrium can impair implantation.
- Diagnosis: Diagnoses like endometriosis, PCOS (polycystic ovary syndrome), or immune-related factors can affect receptivity.
- Number of prior transfers: Repeated implantation failure may indicate a need for additional investigation or protocol changes.
- Transfer technique and timing: Physician skill, ultrasound guidance, and endometrial synchronization all contribute to outcomes.
After a transfer, patients are typically advised to monitor for early pregnancy signs and schedule hCG testing, the hormone measured in a pregnancy test, approximately ten to fourteen days following the procedure.
Having now discussed evidence-based outcomes, let’s move from data back to practical questions—what should patients be asking and considering on their fertility journey?
Personalizing your path: Key questions to ask and next steps
Knowing the statistics is useful, but knowing the right questions to ask your care team is more powerful. The CDC’s own guidance underscores that national averages are a starting point, not a prediction for any individual patient’s case.
Five essential questions to ask your fertility clinic:
- How do you select which embryos to transfer? Ask about grading criteria, PGT use, and whether the clinic prioritizes single embryo transfer to reduce multiple pregnancy risk.
- What protocols do you use for endometrial preparation? Understand whether your clinic individualizes hormone regimens or applies a standard protocol to all patients.
- How do you handle recurrent implantation failure? If this is not a first attempt, you need to know what additional diagnostics or modifications your team will offer.
- What is your lab’s fertilization and blastocyst development rate? These numbers reflect the quality of the laboratory environment, which directly affects embryo viability before transfer.
- Do you offer individualized IVF options that adapt to my specific diagnosis and cycle response? Cookie-cutter protocols are less effective than those tailored to each patient’s biology.
Transparency in these conversations is not just reassuring. It is clinically meaningful. Patients who understand the rationale behind their treatment plan are better positioned to adhere to protocols, flag concerns early, and make informed decisions about next steps, including whether donor eggs or a gestational carrier may be appropriate options to explore through egg donation.
What most people miss about embryo transfer—and what actually works
At Life IVF Center, we have seen a consistent pattern in how patients approach embryo transfer, and it reveals a gap that national statistics simply cannot address. Most people walk into a consultation focused on clinic averages or a neighbor’s success story. What they underestimate is how profoundly individual biology, communication quality, and protocol personalization shape outcomes.
Patients who engage actively with their care team, who ask specific questions about their endometrial lining, their embryo grading, and their protocol rationale, tend to experience fewer surprises and, importantly, fewer avoidable setbacks. This is not anecdotal. Research on patient engagement in reproductive medicine consistently shows that informed patients have better adherence to medication schedules, more accurate symptom reporting, and stronger therapeutic relationships with their providers.
The hard truth about tailoring IVF protocols is that the “one size fits all” approach still persists at some clinics. It is easier and faster to apply a standard protocol than to analyze each patient’s response history, adjust hormone dosing based on real-time monitoring, and revisit the transfer plan if the endometrium is not responding optimally. But this efficiency often comes at the cost of outcomes.
What actually works is precision, patience, and proactive communication. Knowing when to wait a cycle, when to freeze rather than transfer fresh, and when to investigate recurrent failure rather than simply retry the same protocol separates good IVF care from excellent IVF care. Patients deserve that distinction clearly explained before they begin treatment, not after a second or third failed attempt.
Explore advanced embryo transfer support with Life IVF Center
At Life IVF Center, embryo transfer is never treated as a routine step. Every transfer is approached with individualized preparation, advanced ultrasound guidance, and protocols shaped by each patient’s unique biological profile.
Whether you are exploring IVF for the first time or seeking answers after a prior cycle, Life IVF Center’s Precision IVF® approach offers a framework built around your specific needs. You can start by reviewing intro to IVF and embryo transfer resources to build your foundational knowledge, or explore Precision IVF options to understand how individualized protocols are designed. Our team across multiple Southern California locations is ready to support your journey with the clinical depth and compassionate care you deserve.
Frequently asked questions
Is embryo transfer painful?
Most patients experience only mild discomfort during embryo transfer, such as light cramping, as anesthesia is generally not required for this procedure.
How long after embryo transfer does implantation occur?
Implantation typically occurs within one to five days following the embryo transfer procedure, depending on the embryo’s developmental stage at the time of transfer.
What affects my chances of success with embryo transfer?
Age, cause of infertility, embryo quality, and uterine preparation are key influencing factors, and outcomes vary by age and diagnosis according to CDC-reported national data.
What is the difference between fresh and frozen embryo transfer?
Fresh embryos are transferred shortly after retrieval, while frozen embryos are thawed and transferred in a later cycle following additional preparation, which is especially important because endometrial preparation is a key nuance in frozen transfers.
Where can I check the latest national IVF and embryo transfer success rates?
You can access current success data directly through the CDC ART Success Rates tool, which provides cumulative live-birth-related measures reported by fertility clinics nationwide.
Recommended
- Fertilization & Culturing – Life IVF Center
- Male Fertility: Tips and Key Factors for IVF Success – Life IVF Center
- Pre-IVF Pricing – Life IVF Center
- Precision IVF: Transforming Fertility Treatment – Life IVF Center